
IS THE SPIKE PROTEIN TRANSFORMING THE VASCULATURE INTO "TUMOR BLOOD VESSELS?"
The Spike Protein, Autopsies and VE-Cadherin: Continuing development of the SPED hypothesis
In 14 out of 16 autopsies for COVID-19 victims, a startling finding was made.
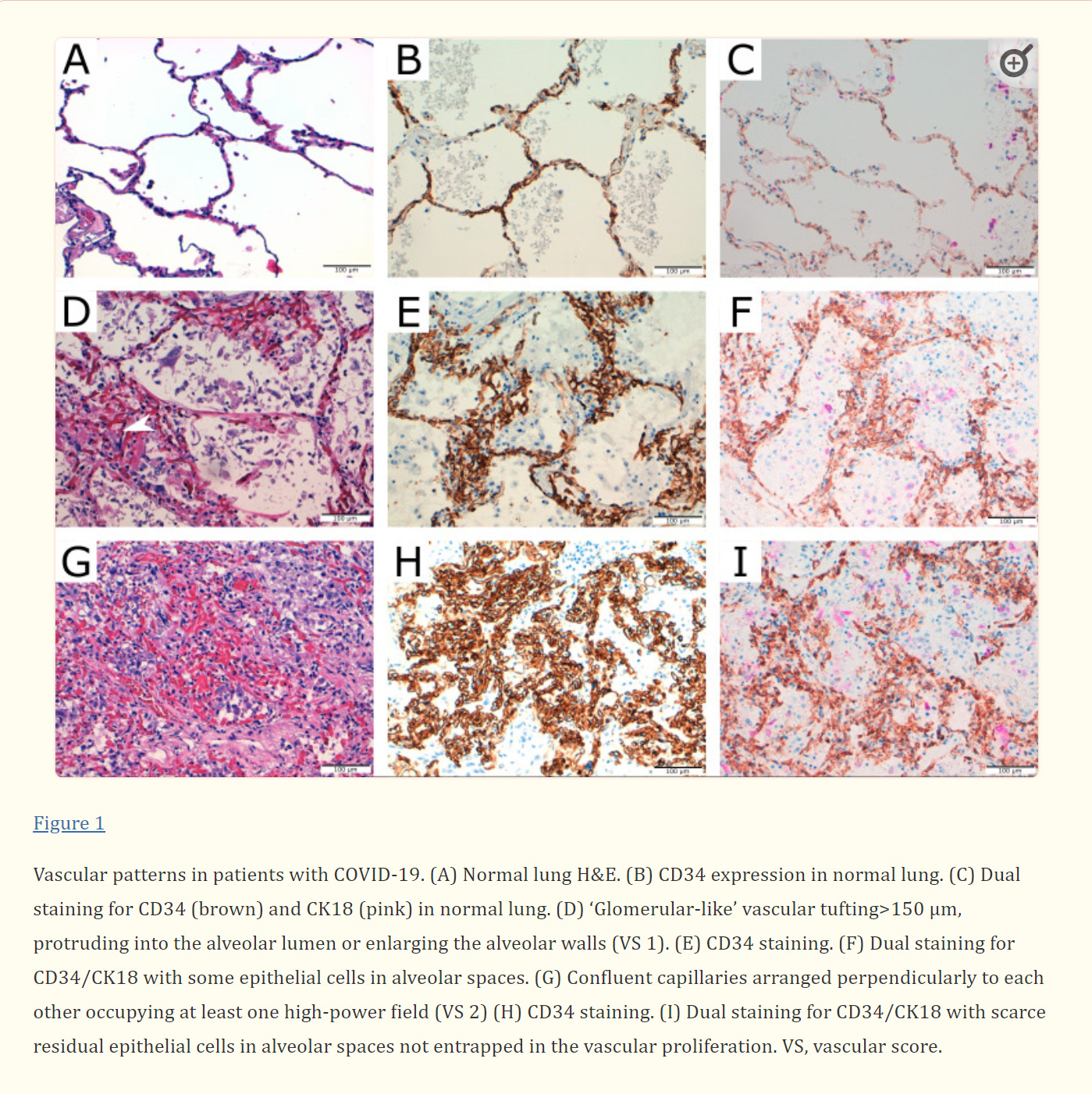
Glomeruloid-like microscopic foci and/or coalescent vascular proliferations measuring up to 2 cm were present in the lung of 14 out of 16 autopsied patients. These lesions expressed CD31, CD34 and vascular endothelial cadherin (VE-Cadherin).
Additionally:
After injury, angiogenesis occurs to restore lung vasculature. Ackermann et al demonstrated that the amount of new vessel growth in the lung of patients with COVID-19 was 2.7 times higher than in the lungs from patients with influenza.
Pulmonary vascular proliferation in patients with severe COVID-19: an autopsy study
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7992389/
I believe this is occurring due to the Spike Protein’s interaction with VE-Cadherin.
Here, we showed that the spike protein of SARS-CoV-2 depends on its RGD motif to drive barrier dysregulation through hijacking integrin αVβ3. This triggers the redistribution and internalization of major junction protein VE-Cadherin which leads to the barrier disruption phenotype.
Vascular dysregulation following SARS-CoV-2 infection involves integrin signalling through a VE-Cadherin mediated pathway
https://www.biorxiv.org/content/10.1101/2022.03.15.484274v1.full
And, what does this mean?
We have the transformation of healthy endothelium into that of tumor endothelium.
In contrast, tumor blood vessels usually have deficient pericyte and VSMC coverage, which are not tightly attached to the ECs. They are often immature, less contractile and their shape is abnormal. The Basement Membrane (BM) is often degraded and loosely attached to the EC, or it is thick and thus hinder contraction and perfusion. The morphology of ECs are changed and cell-cell connections are often lost. As a result, vessels get dilated negatively impacting the ratio of vessel surface, which supplies the tumor tissue with oxygen and nutrients. Tumor cells can press in the instable vessel wall, thus vessel diameter becomes uneven. Because of this and the altered EC morphology, vessels get curved and form serpentine-like structures, and may also create high amount of anastomosis within the vessel wall. This is further supported by the impaired cell junctions, resulting in the loss of the signals of oxygen sensors, eventually leading to abnormal flow in the tumor vasculature. Because of the degraded BM, ECs lose their polarity, thus get detached from the endothelial layer and may form a plug in the lumen resulting in thrombosis and further injury of the vessel wall.
Tumor distribution and efficacy of antiangiogenic receptor tyrosine kinase inhibitors
https://repo.lib.semmelweis.hu/bitstream/handle/123456789/7025/torokszilvia.d_DOIs.pdf?sequence=1
And explains why COVID-91, Long COVID and Spike Protein Endothelial Disease (SPED) are microvascular diseases.
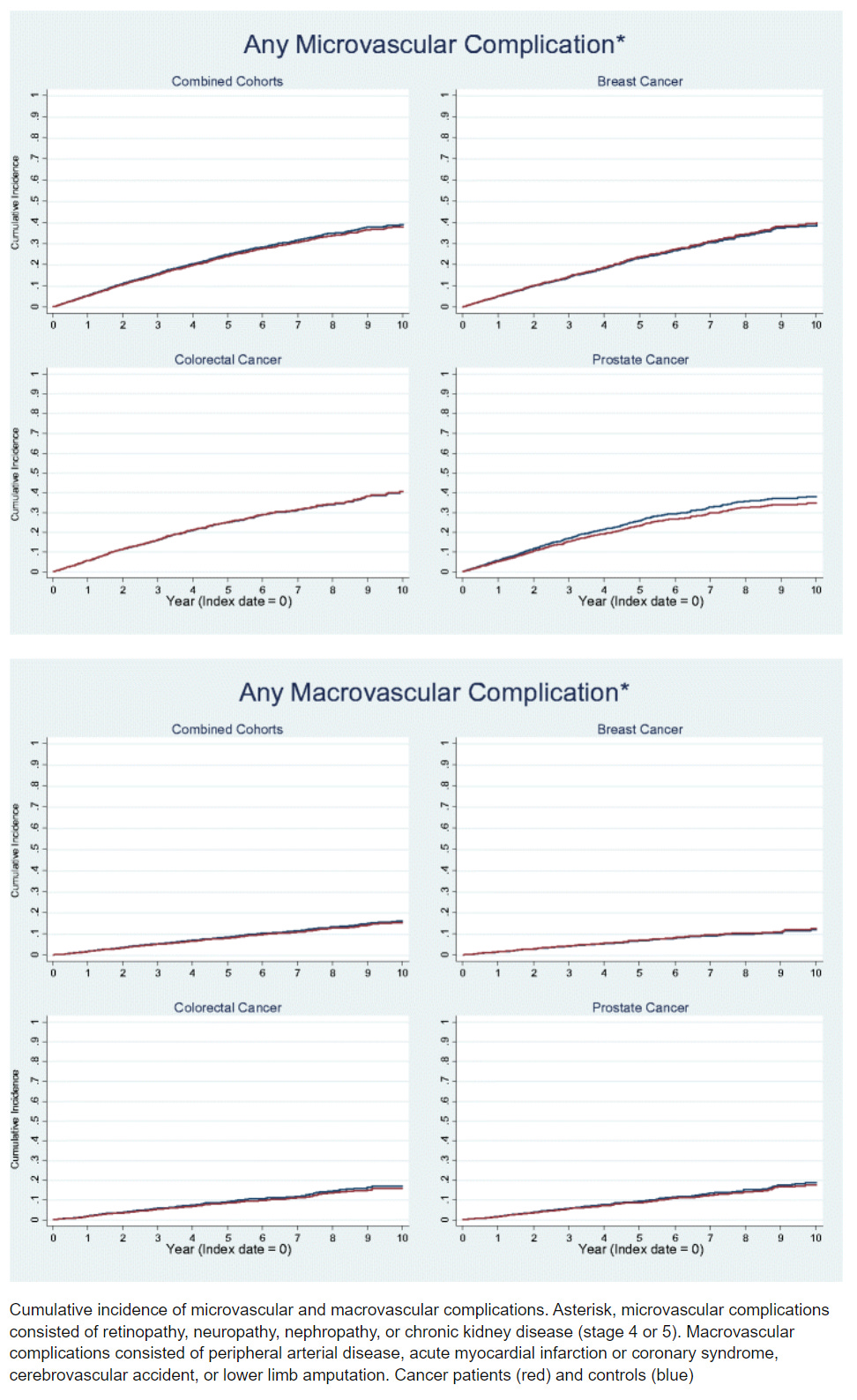
Outcomes of preexisting diabetes mellitus in breast, colorectal, and prostate cancer
https://www.researchgate.net/publication/318640522_Outcomes_of_preexisting_diabetes_mellitus_in_breast_colorectal_and_prostate_cancer
This may all seem rather sci-fi movie fantastical – but it isn’t. This very condition can be induced by using viruses to EXPRESS CERTAIN GENES. And this was back in 2001.
Glomeruloid bodies are a defining histological feature of glioblastoma multiforme and some other tumors and vascular malformations. Little is known about their pathogenesis. We injected a nonreplicating adenoviral vector engineered to express vascular permeability factor/vascular endothelial growth factor-164 (VPF/VEGF164) into the ears of athymic mice. This vector infected local cells that strongly expressed VPF/VEGF164 mRNA for 10 to 14 days, after which expression gradually declined. Locally expressed VPF/VEGF164 induced an early increase in microvascular permeability, leading within 24 hours to edema and deposition of extravascular fibrin; in addition, many pre-existing microvessels enlarged to form thin-walled, pericyte-poor, “mother” vessels. Glomeruloid body precursors were first detected at 3 days as focal accumulations of rapidly proliferating cells in the endothelial lining of mother vessels, immediately adjacent to cells expressing VPF/VEGF164. Initially, glomeruloid bodies were comprised of endothelial cells but subsequently pericytes and macrophages also participated. As they enlarged by endothelial cell and pericyte proliferation, glomeruloid bodies severely compromised mother vessel lumens and blood flow. Subsequently, as VPF/VEGF164 expression declined, glomeruloid bodies devolved throughout a period of weeks by apoptosis and reorganization into normal-appearing microvessels. These results provide the first animal model for inducing glomeruloid bodies and indicate that VPF/VEGF164 is sufficient for their induction and necessary for their maintenance.
Glomeruloid Microvascular Proliferation Follows Adenoviral Vascular Permeability Factor/Vascular Endothelial Growth Factor-164 Gene Delivery
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1850349/
If this is indeed happening, and I believe it is, then this alone would explain the explosion in new and recurring cancers, as dormant microtumors are, like so many zombies, risen from their graves.












Your research continues to startle. Monsters rose from their graves in the form of select administrators and politicians. Hopefully, they get stakes or silver bullets in their chest cavaties (no hearts there.) Thank you again for your work.
Thank you as always, Walter! Your insights have helped to keep our family safe in all this.