Friday Hope: Natalizumab: A Therapeutic for MS May Prove Effective as a Treatment for COVID and Spike Protein-Related Neuropathologies
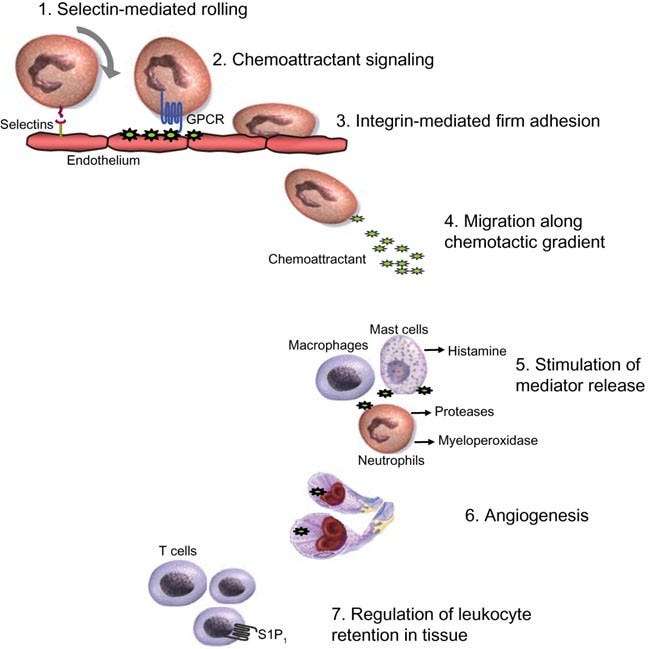
Inhibiting the chemotaxis of immune cells into the intestinal lining and blood-brain-barrier.
Given my recent findings that point towards the Spike Protein of SARS-CoV-2 drawing immune cells into tissues and destroying them, I began a search for a treatment that could ameliorate this. I believe I have found one.
Natalizumab, sold under the brand name Tysabri among others, is a medication used to treat multiple sclerosis and Crohn's disease. It is a humanized monoclonal antibody against the cell adhesion molecule α4-integrin. It is given by intravenous infusion every 28 days. The drug is believed to work by reducing the ability of inflammatory immune cells to attach to and pass through the cell layers lining the intestines and blood–brain barrier. Natalizumab has proven effective in treating the symptoms of both diseases, preventing relapse, vision loss, cognitive decline and significantly improving quality of life in people with multiple sclerosis, as well as increasing rates of remission and preventing relapse in multiple sclerosis.
Natalizumab, is a monoclonal antibody which targets a protein called α4β1 integrin on white blood cells involved in inflammation. By attaching to integrin, natalizumab is thought to stop white blood cells from entering the brain and spinal cord tissue, thereby reducing inflammation and the resulting nerve damage.
Natalizumab
https://en.wikipedia.org/wiki/Natalizumab
Indeed the Spike Protein interacts with α4-integrin, and inhibiting this interaction has proven effective.
As pulmonary epithelial cells represent a prime target for SARS-CoV-2, we have investigated the binding of the spike protein to the human pulmonary epithelial cell lines 11-18 and QG-56. We have found that these epithelial cell lines showed good binding to the spike protein. β1 integrins are the predominant integrins expressed in pulmonary epithelial cells under physiologic conditions, although some levels of αVβ3, α6β4, αVβ5, αVβ6, and αVβ8 integrins are also expressed and upregulated under pathologic conditions such as in various cancers and fibrosis. Thus, we have tested the effects of anti-β1 and anti-αV integrin inhibitory antibodies that block all β1 integrins (α1β1, α2β1, α3β1, α4β1, α5β1, α6β1, α7β1, α8β1, α9β1, α10β1, and α11β1) and αV integrins (αVβ1, αVβ3, αVβ5, αVβ6, and αVβ8), respectively. The adhesive interactions of pulmonary epithelial cells with the spike 1 (S1) protein are potently inhibited by pretreatment with anti-β1 or anti-αV integrin inhibitory antibodies.
The Spike Glycoprotein of SARS-CoV-2 Binds to β1 Integrins Expressed on the Surface of Lung Epithelial Cells
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8069079/
Why I believe Natalizumab (NTZ) should be trialed for COVID, Long COVID and Spike Protein-related Neuropathologies is that MS patients on NTZ did quite well when infected with SARS-CoV-2.
We reported on 6 patients treated with NTZ for relapsing MS during active COVID-19 infection, who recovered without reporting any worsening or new symptoms. Most of the patients were asymptomatic, with the exception of one patient who had a slight worst COVID-19 clinical course. No patients received O2-therapy or required intensive care. No neurological complications were observed.
Natalizumab administration in multiple sclerosis patients during active SARS-CoV-2 infection: a case series
https://bmcneurol.biomedcentral.com/articles/10.1186/s12883-021-02421-3
Natalizumab may help those suffering from long-standing Long COVID and may help those with acute infection by reducing severity of symptoms by preventing the chemotaxis of immune cells into the brain and intestinal lining.



Great write up Walter but I think I will stick to natural remedies...anything manufactured by big pharma make me want to run the other way !
walter - take it more easy. you cant come up with a different solution every friday. do what you feel like and do not feel pressured to find something for friday. this is not easy, and what you are up against has been at works for decades ... no worries. as always, many thanks for all you do!!!