Friday Hope: D-Glucosamine: Inhibits SARS-CoV-2 Replication and Infection
Not highlighted enough in the paper: D-Glucosamine significantly decreased Spike Expression
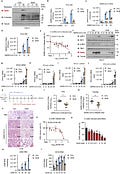
GlcN enhances SARS-CoV-2 induced IFNs signaling and exhibits a broad-spectrum antiviral activity against multiple HCoVs. a Calu-3 cells were treated with GlcN for 3 h at 20 mM, infected with SARS-CoV-2 (MOI = 1) for 4 and 24 h. Immunoblotting of SARS-CoV-2 spike and O-GlcNAcylation level were performed. b Calu-3 cells were treated with GlcN for 3 h at 20 mM, infected with SARS-CoV-2 at an MOI of 0.1 or 0.5. Infectious supernatant was collected at 24 h post infection. Virus titers were measured. c, d Calu-3 cells were treated with GlcN for 3 h at 20 mM, infected with SARS-CoV-2 (MOI = 1) for 6, 16 and 24 h. Analysis of SARS CoV-2 RNA level in cells c, virus titer in supernatant d were performed. e Calu-3 cells were treated with GlcN ranging from 0.1 to 40 mM for 3 h, infected with SARS-CoV-2 (MOI = 1) for 24 h. Cells were stained for the SARS-CoV-2 N protein and imaged. The percentage of SARS-CoV-2 staining was quantified as shown. f–j Calu-3 cells were treated with GlcN for 3 h at 20 mM, infected with SARS-CoV-2 for 6, 16, 24, and 48 h. Immunoblotting of SARS-CoV-2 spike, phosphorylated IRF3 and TBK1 were performed f. qRT-PCR analysis of IFN-β g, IFN-α2 h, IFN-λ1 i, and IFN-λ2/3 j expression was performed. The virus titer of SARS-CoV-2 used in (a, c, d, f–j) was 3.5 × 106 PFU/mL, used in b, e was 1.73 × 106 PFU/mL. k Schematic diagram of the mice experiment. 6 weeks old Balb/c mice were intragastric administration of GlcN (350 mg/kg) for 3 days. Mice were infected with SARS-CoV-2 strain MASCp6 intranasally. Infection for 3 days, mice were sacrificed and tissues were collected. l, m mice were treated as k. qRT-PCR analysis of SARS-CoV-2 RNA level in lung l and trachea m sections were performed. n H&E-stained analysis showing inflammatory cell infiltration (red arrow), alveolar septal thickening (yellow arrow) and fibrin exudation (green arrow) in lungs from mice treated as k. The virus titer of SARS-CoV-2 used in l–n was 4 × 105 PFU/mL. o, p Calu-3 cells were treated with GlcN ranging from 0.1 to 20 mM for 3 h, infected with 229E (1 × 107 PFU/mL) at an MOI of 1 for 24 h. RNA and infectious supernatant were collected. Dose-response curves of GlcN is shown and EC50 is indicated above the curve o. The percentage of 229E infection calculated by viral RNA is shown as indicated p. q, r Calu-3 cells were treated with GlcN for 3 h at 20 mM, infected with 229E or OC43 at an MOI of 1 for 6, 18, 24, 30 h. qRT-PCR analysis of 229E q and OC43 r RNA expression was performed. qRT-PCR results are presented relative to those of GAPDH g–j. Data were shown as means ± SEM l, m, means ± SD b–e, g–j, o–r from triplicates (biological replicates), *p < 0.05, **p < 0.01, and ***p < 0.001 (two-tailed Student’s t test)
I believe this is an important finding which not only may prevent infection by/severity of COVID, but may also dramatically reduce the expression of Spike Protein by our cells.
A Letter to the Editor was published on Tuesday which showed that a very common, safe and readily available supplement was very powerful in inhibiting SARS-CoV-2 replication. It does this by increasing the expression of the ever-important Interferon. As readers may be aware, it has readily been proven that defects in Interferon response are a critical factor in the development of severe COVID.
Let us look at the mechanisms of how D-Glucosamine inhibits SARS-CoV-2 replication.
We recently identified that O-GlcNAcylation, a posttranslational modification derived from hexosamine biosynthetic pathway (HBP), is essential for virus-induced MAVS activation and IFN signaling. We demonstrated that D-glucosamine (GlcN), a commonly used dietary supplement, increases MAVS O-GlcNAcylation and enhances MAVS-mediated IFN signaling, and thereby exhibits a broad-spectrum antiviral activity.6 In this communication, we explore the potential broad-spectrum antiviral activity of GlcN against HCoVs. We first established SARS-CoV-2 infection model using human lung epithelial cell line Calu-3 and human liver cancer cell line Huh7, respectively. Calu-3 or Huh7 cells were treated with GlcN (at a final concentration of 20 mM) for 3 h, infected with SARS-CoV-2 at an multiplicity of infection (MOI) of 1. Upon infection for 24 h, we observed that SARS-CoV-2 infected cells showed a significantly enhanced intensity of cellular O-GlcNAcylation comparing to the non-infected group (Fig. 1a, Supplementary Fig. 1), indicating SARS-CoV-2 promotes HBP metabolism and protein O-GlcNAcylation in host cells. This is akin to our previous observation in RNA virus infection including IAV and VSV.6 As expected, we found that GlcN significantly increased the cellular level of O-GlcNAcylation and substantially suppressed SARS-CoV-2 replication in infected lung epithelial cells as measured by SARS-CoV-2 spike protein expression (Fig. 1a). Virus titers in the supernatant were significantly reduced (P < 0.01) in GlcN treatment group (Fig. 1b). Time course measurements (6 to 24 h post infection) further confirmed our observation, GlcN treatment significantly inhibited replication of SARS-CoV-2, consistent with titer reduction (Fig. 1c, d).
As far as safety is concerned, the authors noticed no cytotoxicity regardless of dose of D-Glucosamine given. At the same time, it was observed that D-Glucosamine also inhibits INFECTION.
The fraction of infected cells was quantified. GlcN inhibited SARS-CoV-2 infection in Calu-3 cells with a EC50 value of 11.82 mM (Fig. 1e). The cytotoxicity of GlcN was examined in parallel with antiviral activity. We did not observe any measurable cytotoxicity at all concentrations (Supplementary Fig. 1c). These results suggest that GlcN effectively inhibits SARS-CoV-2 replication and infection in vitro.
However, a finding the authors gave but one sentence to, I believe could be an indication that D-Glucosamine may help those with Long COVID and Spike Protein induced afflictions.
In an effort to better understand the antiviral mechanism of GlcN against SARS-CoV-2, we examined the SARS-CoV-2 induced interferon response in the presence of GlcN. We observed that GlcN treatment resulted in a considerable increase of both phosphorylated IRF3 and phosphorylated TBK1, and thereby promoted IFN signaling in response to SARS-CoV-2. This in turn decreased the expression of SARS-CoV-2 spike protein in GlcN treatment group (Fig. 1f).
Clearly, additional studies are needed, and I would discuss these findings with your PCP. I will personally be adding GlcN to my regimen.
Regarding dosing (again, not medical advice, discuss any supplement with your PCP), I will leave the final words of this post to the study’s authors.
Overall, our work demonstrated that GlcN shows potential efficacy against multiple HCoVs including SARS-CoV-2, 229E and OC43 in both cell-based and mouse infection model. GlcN has been clinically applied for the treatment of osteoarthritis for more than 50 years. As a nutrient supplement, orally administrated GlcN at daily-doses ranging from 750 to 3500 mg is well tolerated in human subjects. Given the safety profile and its broad-spectrum anti-HCoVs activity, GlcN may serve as a promising drug for preventing the spread of SARS-CoV-2 and its emerging variants in healthy populations.
Oral administration of D-glucosamine confers broad-spectrum protection against human coronaviruses including SARS-CoV-2
https://www.nature.com/articles/s41392-023-01483-8
Please have a healthy, happy and blessed Juneteenth weekend.


Thank you for continuing to research this; I hope the people that I forward your content to actually subscribe. I think you are benefiting the people who need help healing from the injections. Blessings to you!!!
Wow! Excellent news! Thank you Walter! Peace.