Friday Hope: Cinnamon: Cinnamaldehyde as Adjuvant Therapy for COVID/Spike Protein Inflammation
Reduces levels of IL-1β and IL-6. Effective as pretreatment and during active infection. Also reduces viral replication.
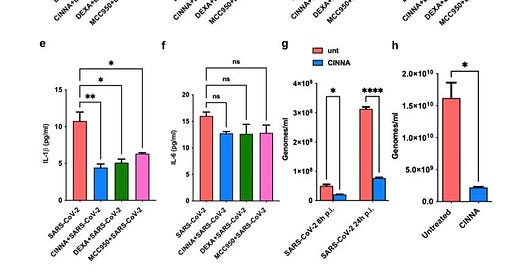
Figure 3. Effects of cinnamaldehyde on SARS-CoV-2 induced inflammation and viral replication. a) Bar graph showing IL-1β levels released by PBMCs of COVID-19 patients (n=13) after LPS-BzATP stimulation and the pretreatment with different compounds: cinnamaldehyde (CINNA), dexamethasone (DEXA) and MCC950. b) Bar graph showing IL-6 levels released by PBMCs of COVID-19 patients (n=13) after LPS-BzATP stimulation and the pretreatment with different compounds: CINNA, DEXA, and MCC950. c) Bar graph showing IL-1β levels released by THP-1 after LPS-BzATP stimulation and the pretreatment with different compounds: cinnamaldehyde (CINNA), dexamethasone (DEXA) and MCC950 (n=3). d) Bar graph showing IL-6 levels released by THP-1 after LPS-BzATP stimulation and the pretreatment with different compounds: CINNA, DEXA, and MCC950 (n=3). e) Bar graph showing IL-1β levels released by THP-1 after SARS-CoV-2 infection (n=6) and the pretreatment with different compounds (n=4): CINNA, DEXA, and MCC950. f) Bar graph showing IL-6 levels released by THP-1 after SARS-CoV-2 infection (n=2) and the pretreatment with different compounds: CINNA (n=4), DEXA (n=2), and MCC950 (n=2). g) Bar graph showing SARS-CoV-2 genomes in CaLu-3 cells 8 and 24 post-infection (h p.i.) with or without the pretreatment with CINNA (n=2). h) Histograms showing SARS-CoV-2 replication rate in VeroE6 cells 48 hours from infection with or without the pretreatment with CINNA (n=2). ns= not significant, *= p<0,05, **= p<0,01, ****= p<0,0001
I have been looking for ways to help reduce inflammation caused by SARS-CoV-2 and its Spike Protein. A very promising paper was published online Tuesday which shows the benefits of cinnamaldehyde (the active compound of cinnamon). The paper showed that levels of IL-1β were reduced dramatically by cinnamaldehyde and levels of IL-6 were somewhat attenuated.
We identified the natural compound cinnamaldehyde (CINNA) as a promising candidate since it has been shown to act on NLRP-3 inflammasome reducing IL-1β release. At the same time, we tested the effects of dexamethasone on these pathways, which nowadays represents one of the standard treatments for severe COVID-19. Also, the known NLRP3 inhibitor MCC950 was used as a control. Notably, CINNA pretreatment strongly decreased LPS + BzATP-induced inflammation in COVID-19 patients’ PBMCs, by inhibiting SARS-CoV-2 enhanced proinflammatory state (Fig. 3a, b). The effects of CINNA were stronger even compared with dexamethasone and MCC950, which were both ineffective in diminishing IL-6 release (Fig. 3b). We then tested the direct effect of CINNA pretreatment on LPS-induced inflammation and SARS-CoV-2 infection on THP-1 cells. Interestingly, in this scenario, CINNA strongly reduced IL-1β and IL-6 secretion induced by LPS as well as dexamethasone and MCC950 (Fig. 3c, d) as well known from literature. However, after viral infection, CINNA could reduce IL-1β levels but not significantly counteract the IL-6 one.
What I find very interesting about this research is that PRETREATMENT with cinnamaldehyde more effectively reduced IL-1β and IL-6 levels than AFTER SARS-CoV-2 infection, which found that only IL-1β was significantly reduced.
Furthermore, NASAL ADMINISTRATION of cinnamaldehyde during infection was very effective at reducing IL-1B levels.
We tested the anti-inflammatory capacity of CINNA, giving it by aerosol as pretreatment before LPS intranasal injection (CINNA + LPS). As a control, we performed aerosol administration of DEXA (DEXA + LPS). As expected, LPS treatment (LPS) was able to increase IL-1β release compared to the control group (CTRL), while both treatments with CINNA and DEXA exhibited a large inhibitory effect on the levels of IL-1β release induced by intranasal LPS administration.
There is an added benefit of cinnamaldehyde (CINNA) reducing viral replication as well. This is clearly another reason to use cinnamon as a prophylactic. Especially as it de facto then reduces the expression of Spike Protein.
In addition, our data proved that CINNA pre-treatment reduces the viral replication rate in lung cells, suggesting that CINNA might provide beneficial effects by down-regulating the synthesis of IL-1β also by reducing viral spreading.
The paper concludes that this is not only likely to be effective with SARS-CoV-2 infection, but other respiratory infections, as well.
Inflammation is not an exclusive characteristic of COVID-19 but is also present in many other forms of pneumonia caused by various pathogens. Therefore, its application could also be relevant in the treatment of different types of pneumonia. Hence, CINNA represents a promising dietary supplement to reduce the pro-inflammatory effects due to pathogens-induced pulmonary infections.
SARS-CoV-2 infection as a model to study the effect of cinnamaldehyde as adjuvant therapy for viral pneumonia.
https://www.researchsquare.com/article/rs-3072092/v1
If we concentrate on a regiment to limit exposure to/expression of Spike Protein, and limit both acute and chronic inflammation, we may be able to avoid some of the more deleterious consequences of the SARS-CoV-2 virus and its Spike Protein.
I wish all in the USA a blessed Independence Day weekend, and a blessed weekend to all. Thank you for your continued support.



It appears various spices, herbs and plants, such as Cinnamon, Turmeric, Dandelion, Licorice, Fennel Seed, Pine Needles, etc, are just waiting to save our lives.
Interesting. We have a family recipe been around forever that we always have made when people are sick with cold , flu etc. A mixture of apricot nectar, pineapple juice , apple cider ( not juice), orange juice and cinnamon stix ( 3 whole) and 12 whole cloves simmered. The steam from it is great andthe warm drink taste good and always seems to help esp with chest congestion. I make it for friends as well.