Discussing A Recent Preprint from Yale: Is Post-Vaccination Syndrome (PVS) Actually a New Spike-Induced Amyloidosis?
When you break down the symptoms of Post-Vaccination Syndrome, a very strong case can be made for Post-Vaccination Amyloidosis.
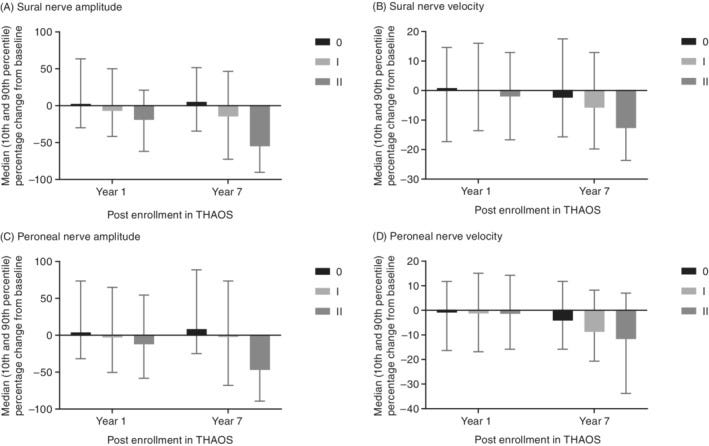
Median (10th and 90th percentile) percentage change from baseline in sural and peroneal nerve conduction by latest mPND score in patients with ATTRv amyloidosis and nerve conduction studies. mPND score IIIa, IIIb, and IV values are either not available (no patients with data) or not shown due to low n (n = 1). Data from year 8 not shown due to small number of patients in certain subgroups. ATTRv, hereditary transthyretin amyloidosis; mPND, modified polyneuropathy disability; THAOS, Transthyretin Amyloidosis Outcomes Survey
A preprint appeared on Friday from the Yale Listen to Immune, Symptom and Treatment Experiences Now (LISTEN) study. This preprint focused on what has been called Post-Vaccination Syndrome. The symptoms of this syndrome are described as follows:
The median time from the day of index vaccination to symptom onset was three days (IQR: 1 day to 8 days). The time from vaccination to symptom survey completion was 595 days (IQR: 417 to 661 days). The median Euro-QoL visual analogue scale score was 50 (IQR: 39 to 70). The five most common symptoms were exercise intolerance (71%), excessive fatigue (69%), numbness (63%), brain fog (63%), and neuropathy (63%). In the week before survey completion, participants reported feeling unease (93%), fearfulness (82%), and overwhelmed by worries (81%), as well as feelings of helplessness (80%), anxiety (76%), depression (76%), hopelessness (72%), and worthlessness (49%) at least once.
The cause of this syndrome remains undefined.
A less well-characterized adverse event is a chronic syndrome with symptoms that begin soon after vaccination. A recent preprint from the US National Institutes of Health described 23 people who reported neuropathic symptoms starting within 21 days after vaccination. The cause of this syndrome is undefined, diagnostic tests and evidence-based interventions are lacking, and its connection with vaccination remains controversial.
The authors very carefully word the study to protect the mRNA vaccines and would like us to believe that this is a rare syndrome.
PVS could be caused by several potential mechanisms, including a mechanism related to the vaccination or manufacturing process. It may represent a rare response to vaccines in susceptible individuals.
Post-Vaccination Syndrome: A Descriptive Analysis of Reported Symptoms and Patient Experiences After Covid-19 Immunization
https://www.medrxiv.org/content/10.1101/2023.11.09.23298266v1.full.pdf+html
I will also be referencing another paper which discusses postural orthostatic tachycardia syndrome-like symptoms following COVID-19 vaccination, which includes neuropathy. This second paper dashes the previous authors’ claim that PVS is a “rare” event.
A recently posted preprint showed that following COVID-19 vaccination, a third of the patients who underwent skin biopsy had subthreshold nerve fibre density, thereby fulfilling the pathological criteria for new onset small fibre neuropathy.
What both papers fail to examine is the very likely possibility that the post-vaccination syndromes are caused by a de novo Spike Protein induced amyloidosis.
First, it is imperative to remember that the Spike itself is amyloidogenic.
Our data propose a molecular mechanism for potential amyloidogenesis of SARS-CoV-2 S-protein in humans facilitated by endoproteolysis. The prospective of S-protein amyloidogenesis in COVID-19 disease associated pathogenesis can be important in understanding the disease and long COVID-19.
Amyloidogenesis of SARS-CoV-2 Spike Protein
https://pubs.acs.org/doi/10.1021/jacs.2c03925
I will now demonstrate that, given the amyloidogenic properties of the Spike Protein, a very strong case can be made that these post-vaccinations syndromes are actually caused by a new kind of Spike Protein induced amyloidosis. Let us begin with the observed neuropathy.
Peripheral neuropathy is a common complication of many of the systemic amyloidoses. Although the cause of neuropathy is not entirely clear, it is likely related to amyloid deposition within the nerve. This may lead to focal, multifocal, or diffuse neuropathies involving sensory, motor and/or autonomic fibers. The presenting symptoms depend on the distribution of nerves affected. One of the most common phenotypes is sensorimotor polyneuropathy, which is characterized by symptoms of neuropathic pain, numbness, and in advanced cases weakness.
AMYLOID NEUROPATHIES
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3531896/
Exercise intolerance is present in many forms of amyloidosis. I believe this is highly relevant as the Spike Protein is known to attack the endothelium.
Anoctamin 5 and dysferlin mutations can result in myopathies with similar clinical phenotype. Amyloid deposits can occur in the muscle of patients with dysferlinopathy. We describe a 53-year-old woman with exercise intolerance since childhood, recurrent rhabdomyolysis and late-onset weakness. Muscle biopsy showed amyloid deposits within the blood vessel walls and around muscle fibers.
Amyloidosis and exercise intolerance in ANO5 muscular dystrophy
https://www.nmd-journal.com/article/S0960-8966(11)01309-5/pdf
If amyloid deposits in the valves in the heart, this can lead to leaky (regurgitant) or narrowed (stenotic) valvular disease. Specifically, ATTR amyloidosis has been found in some patients being treated for severe aortic stenosis. Symptoms include shortness of breath, exercise intolerance, lightheadedness and dizziness.
Cardiac Amyloidosis
https://www.hopkinsmedicine.org/health/conditions-and-diseases/cardiac-amyloidosis
Furthermore, the second referenced paper provides ample evidence of the presence of amyloidosis.
A recently posted preprint showed that following COVID-19 vaccination, a third of the patients who underwent skin biopsy had subthreshold nerve fibre density, thereby fulfilling the pathological criteria for new onset small fibre neuropathy . Other study patients had demonstrated either borderline low density or axonal swelling with reduced conduction velocities in distal leg. The authors also reported increased C4d deposition on endothelial cells in skin biopsy samples and the presence of oligoclonal bands in cerebrospinal fluid (CSF).
Postural orthostatic tachycardia syndrome-like symptoms following COVID-19 vaccination: An overview of clinical literature
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10357168/
Let us examine these observations.
C4d Deposition
Cutaneous amyloidosis (CA) is defined by the accumulation of amyloid in the dermis; it might be primary or secondary. The diagnosis is based on histopathological findings with the demonstration of amyloid deposits, confirmed by Congo red stain under the polarized light. Studies on other diagnostic markers are ongoing in the literature. The aim of this study was to demonstrate the utility of C4d staining in the recognition of amyloid in CA and using it as an alternative or substitute marker for the diagnosis. In this retrospective study, 199 skin biopsies with a clinical provisional diagnosis of CA were analyzed, the Congo red stain was performed, and, in a subgroup (n = 97) with histopathological findings probably for CA, C4d immunohistochemistry was assessed. Forty-eight cases of CA were detected. Congo red birefringence was positive in all cases, whereas in 14 cases, it was faded. In these 14 cases, the diagnosis of CA was made by means of Congo red fluorescence and Thioflavin T because the histopathological findings were highly suggestive for CA. All CA cases were positive with C4d, and in 12 of the 49 inflammatory dermatoses, C4d was positive. The interpretation of C4d immunohistochemistry can be performed more easily and rapidly than Congo red evaluation. The sensitivity and specificity of C4d were 100% and 75.5%, respectively. In our experience, C4d staining was a useful method for detecting amyloid deposits in CA. Although Congo red staining is the gold standard for amyloid detection, we propose C4d immunohistochemistry as a routine screening method or hybrid transition while further investigations are completed.
C4d as a Practical Marker for Cutaneous Amyloidosis
https://journals.lww.com/amjdermatopathology/abstract/2022/01000/c4d_as_a_practical_marker_for_cutaneous.5.aspx
Reduced Conduction Velocities
Sensory nerve conduction amplitudes can be absent or reduced. Motor nerve conduction amplitudes can be reduced or normal, with normal to mildly slowed conduction velocities.
AMYLOID NEUROPATHIES
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3531896/
Small Fiber Neuropathy
Neuropathy can be an early manifestation of systemic amyloidosis. Such patients often present with a painful distal and symmetric neuropathy and autonomic symptoms. It can begin as a small fiber neuropathy and progress to involve the large sensory and motor axons (Yang et al, 2010).
Neuropathy and Amyloidosis
https://therapath.com/neuropathy-and-amyloidosis
Perhaps most importantly, we have a cause in amyloidosis for the observed psychological impact of PVS (PVA).
Conclusions: Many patients with AL amyloidosis suffer from depression, anxiety and functional limitations. Psychiatric assessment and treatment is important, and further research is needed to clarify the long-term effects of depression and anxiety in AL amyloidosis.
Depression and anxiety in patients with AL amyloidosis as assessed by the SF-36 questionnaire: experience in 1226 patients
https://pubmed.ncbi.nlm.nih.gov/27460276/
I believe a rethinking of PVS is needed. Further testing, studies and investigation is needed to determine if a new form of Spike Protein induced amyloidosis is the main cause, or a major cause of what has been called Post Vaccination Syndrome.
Thank you for your readership and support.


Funny what an honest, talented researcher can run across when he’s not hindered by institutional diktats and human cowardice :)
DR SYED HAIDER SUBSTACK
Is There a Test for Spike Protein Toxicity?
99.9% of doctors will tell you no, even those who specialize in treating it, but they're dead wrong and it could kill you
https://blog.mygotodoc.com/p/is-there-a-test-for-spike-protein
ARCHIVED ⬇️
https://archive.md/2023.10.24-003652/https://blog.mygotodoc.com/p/is-there-a-test-for-spike-protein