A SUGAR-DRIVEN ENDOTHELIAL DISEASE: The Spike Protein, Glycolysis and Methylglyoxal: Endothelial Damage and Cytokine Storms: Why Metformin and Ketogenic Diets Work
The Spike Protein Increases Glycolytic Metabolism Which I Believe Results in Massive Levels of Methylglyoxal Explaining Endothelial Damage and Cytokine Storms
The Spike Protein of SARS-CoV-2 has been shown to increase (in a DOSE-DEPENDENT MANNER) glycolytic metabolism in monocytes.
A hallmark of COVID-19 is a hyperinflammatory state associated with severity. Monocytes undergo metabolic reprogramming and produce inflammatory cytokines when stimulated with SARS-CoV-2. We hypothesized that binding by the viral spike protein mediates this effect, and that drugs which regulate immunometabolism could inhibit the inflammatory response. Monocytes stimulated with recombinant SARS-CoV-2 spike protein subunit 1 showed a dose-dependent increase in glycolytic metabolism associated with production of pro-inflammatory cytokines.
Metformin Suppresses Monocyte Immunometabolic Activation by SARS-CoV-2 Spike Protein Subunit 1
https://www.frontiersin.org/articles/10.3389/fimmu.2021.733921/full
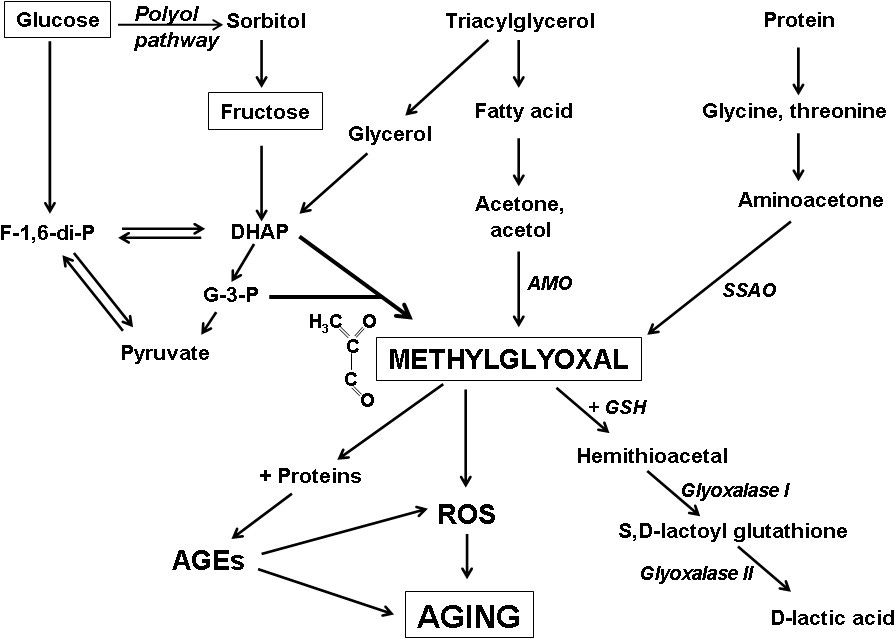
A metabolite of glycolysis, the metabolic process which the Spike Protein increases is Methylglyoxal (MG).
Methylglyoxal (MG) is a side-product of mainly glycolysis that together with reactive oxygen species (ROS) leads to cellular dysregulation in age-related diseases and is a potential causal factor behind diabetes.
Methylglyoxal (MG) is a ubiquitous metabolite that spontaneously reacts with biopolymers forming advanced glycation end-products (AGEs). AGEs are strongly associated with aging-related diseases, including cancer, neurodegenerative diseases, and diabetes.
Methylglyoxal Metabolism and Aging-Related Disease: Moving from Correlation toward Causation
https://www.cell.com/trends/endocrinology-metabolism/fulltext/S1043-2760(19)30206-1
Also, elevated plasma levels of Methylglyoxal were found to be directly correlated with mortality in COVID ICU patients:
Here we found plasma levels of the glycolysis byproduct methylglyoxal (MG) were 4.4-fold higher in ICU patients upon admission that later died (n = 33), and 1.7-fold higher in ICU patients that survived (n = 32), compared to uninfected controls (n = 30). The increased MG in patients that died correlated inversely with the levels of the MG-degrading enzyme glyoxalase-1 (r2 = − 0.50), and its co-factor glutathione (r2 = − 0.63), and positively with monocytes (r2 = 0.29).
Elevated plasma level of the glycolysis byproduct methylglyoxal on admission is an independent biomarker of mortality in ICU COVID-19 patients
https://www.nature.com/articles/s41598-022-12751-y
Why I believe this is particularly important in relation to COVID, Long COVID and what I have called Spike Protein Endothelial Disease (SPED) is that Methylglyoxal itself destroys the Endothelium.
In vitro, MG and TNF-α were shown to synergize in promoting endothelial cell death, which was associated with increased angiopoietin 2 expression and reduced Bcl-2 expression. These results suggest that MG in diabetes increases inflammation, leading to endothelial cell loss.
Methylglyoxal-Induced Endothelial Cell Loss and Inflammation Contribute to the Development of Diabetic Cardiomyopathy
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4878427/
Clearly, it is especially relevant as the Spike Protein itself induces the production of TNF-α.
Modulation of TNF-alpha-converting enzyme by the spike protein of SARS-CoV and ACE2 induces TNF-alpha production and facilitates viral entry
https://pubmed.ncbi.nlm.nih.gov/18490652/
This then brings us to a deeper understanding of why Metformin and Ketogenic diets show a beneficial effect with COVID and SPED.
Thus, in addition to lowering hepatic gluconeogenesis, metformin also scavenges the highly reactive MG in vivo, thereby reducing potentially detrimental MG protein adducts, with subsequent reductions in diabetic complications.
Metformin Scavenges Methylglyoxal To Form a Novel Imidazolinone Metabolite in Humans
https://pubs.acs.org/doi/10.1021/acs.chemrestox.5b00497
In summary, a ketogenic diet prevented the onset of methylglyoxal-evoked mechanical allodynia and increased scavenging of circulating methylglyoxal.
A Ketogenic Diet Prevents Methylglyoxal-Evoked Nociception by Scavenging Methylglyoxal
https://www.sciencedirect.com/science/article/abs/pii/S1526590022001420
Indeed, the Spike Protein’s engine of increased glycolysis (therefore increased Methyglyoxal) and TNF-α production may be a significant factor in the observed emergence of vastly increased age-related diseases.
An excess of MG formation can increase ROS production and cause oxidative stress. MG reacts with proteins, DNA and other biomolecules, and is a major precursor of advanced glycation end products (AGEs). AGEs are also associated with the aging process and age-related diseases such as cardiovascular complications of diabetes, neurodegenerative diseases and connective tissue disorders.
Oxidative stress and aging: Is methylglyoxal the hidden enemy?
https://cdnsciencepub.com/doi/10.1139/Y10-001



Thank you, once again, Walter. I pray you have a Happy New Year!
I read that the supplement Berberine is similar to Metformin.