Friday Hope: Abundant Natural Amyloid Inhibitors: A Review Article
We begin to see a picture emerging of certain key players demonstrating multiple benefits in combating SARS-CoV-2 and its Spike Protein.
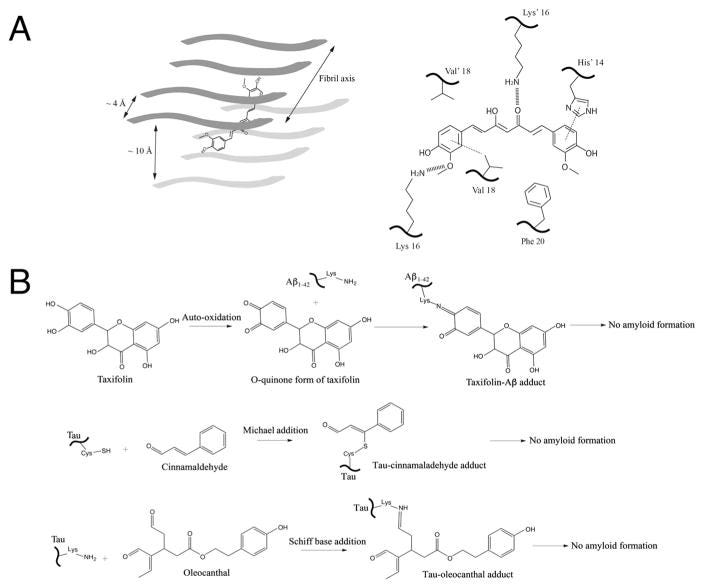
Schematic representations of several proposed mechanisms between inhibitors and amyloid proteins. (A) Non-covalent interaction mechanisms with curcumin as an example. Left panel: The planar curcumin molecule is depicted by a cartoon schematic within the cross beta spine of an octomeric fibrillar backbone. This representation is based on the structural model of curcumin bound to the VQIVYK segment from the tau protein as well as MD simulation results of curcumin docking onto Aβ hexapeptide KLVFFA [30]. Right panel: key non-covalent interactions occur within the cross beta spine of full-length amyloid β peptide and curcumin, as depicted from a recent MD simulation study [32]. His14 undergoes π–π stacking (dotted line) with one end of the aromatic heads of curcumin, which is also positioned in a hydrophobic area near Phe20 (bottom right). The central keto-enol functional groups and as well as the aromatic head (bottom left) undergo hydrogen bonding with lysine residues located on opposite sides of the cross beta spine (hashed lines). Additionally, π–alkyl interactions (depicted by dotted line) were seen between the aromatic head of curcumin (bottom left) and Val18 residues. (B) Covalent interaction mechanisms. Small molecule natural compounds containing electrophilic functional groups such as o-quinones and aldehydes form covalent adducts with amyloidogenic proteins and prevent amyloid formation. (Top panel) Taxifolin forms covalent adducts with the side chain amine group of lysine in Aβ1-42 via Schiff base formation via o-quinone intermediates. (Middle and Lower panels) Tau is covalently modified by the aldehyde functional groups in the case of cinnamaldehyde (middle panel) and oleocanthal (lower panel) via Michael addition and Schiff base respectively. Such conjugation prevents protein amyloid growth and formation.
Readers of this Substack are by now quite aware of the amyloidogenic properties of SARS-CoV-2's Spike Protein. These properties have been brought, once again, to the fore by the discovery of amyloid aggregates in the blood products of those who received mRNA injections. Please see my previous Substack for details.
There are many diseases that involve amyloidosis. From Alzheimer’s to Diabetes and far beyond. Though these diseases affect very different organs and systems, two fundamental principles must apply in dealing with them. Inhibition of amyloid formation, and degradation of those that do form.
One of the current strategies aimed at identifying therapeutic lead compounds for amyloidosis focuses on inhibiting amyloid aggregation by (i) inhibiting toxic amyloid formation and/or stabilizing its native form from aggregating and (ii) remodeling or degrading toxic amyloid oligomers and/or insoluble fibrils.
One aspect of Spike Protein therapeutics that emerges from our study is that many natural compounds have multiple benefits in treating not only Spike Protein pathologies but also acute COVID, itself. Here, we delve into two familiar friends: Curcumin and EGCG.
CURCUMIN
Curcumin has been documented to modulate amyloid assembly in various amyloid systems. Because curcumin has been identified as a potential pan-assay interference compound (PAINS) [61], it is especially important to have multiple orthogonal assays to validate the bioactivities of curcumin. Nonetheless, extensive literature shows that curcumin prevents amyloid formation, amyloid induced cytotoxicity, and provides beneficial in vivo effects including reduced plaque burden (for a recent review, see [62]; Table 1) via (i) inhibiting amyloid aggregation in the instances of amylin or Aβ [60,63–65] (ii) Accelerating α-synuclein aggregation that results in less toxic intermediates and insoluble aggregates [59,66].
For those interested in the proposed mechanism of Curcumin in inhibiting amyloids, I offer the following:
A common phenomenon observed in each study was that curcumin intercalated within the hydrophobic core of all amyloid assemblies regardless of the type of peptide or the quaternary structure of each complex. The non-covalent interactions observed within these complexes give not only mechanistic insights into how curcumin exerts its anti-amyloid effects [58,59,65], but also show how the molecular scaffold of curcumin is capable of targeting the cross beta spine present in all amyloid fibrillar structures, as well as α-helical oligomers that may play an important role during the early events of amyloid aggregation [67,68].
EGCG
Not only does EGCG appear to inhibit amyloid formation, it is also shown to remodel (degrade) existing amyloid fibrils.
EGCG exerts powerful anti-amyloid effects against a number of amyloidogenic proteins. It has the ability to prevent the formation of toxic prefibrillar oligomers (while stabilizing non-toxic off pathway oligomers), as well as inhibit amyloid fibril formation and remodel previously existing amyloid fibrils into less toxic insoluble aggregates [47,50,52,57,74–76]. Numerous studies have elucidated some of the key non-covalent binding events and interactions that mediate these effects. Multiple studies suggest that EGCG undergoes non-specific hydrophobic and hydrogen bonding interactions that can mediate its anti-amyloid activities: (i) Nitro blue tetrazolium (NBT) dye staining analysis as well as NMR data suggest that EGCG can bind to natively unstructured α-synuclein and amyloid β peptide or denatured bovine serum albumin but not other native globular proteins [57]. These data suggest that EGCG may have a propensity to target unfolded or natively unstructured proteins, presumably via non-specific backbone interactions [57]. (ii) EGCG can remodel preformed amyloid generated by the mutant form of acetylated fragment of yeast prion protein Sup35 (GNNQQNFQQF) but not the native fragment (GNNQQYQQY). Such differential effects may be due to the mutant fragment possessing more hydrophobic binding regions that can interact with EGCG [47]. (iii) Using a series of amylin mutants, Raleigh’s group investigated the importance of residue-specific aromatic/hydrophobic or covalent interactions that may mediate EGCG-induced amyloid inhibition and/or remodeling activities. They concluded that neither was critical, and that backbone hydrogen bonding/hydrophobic interactions likely mediate the effects of EGCG [49]. Other studies indicate that the ability of EGCG to inhibit amylin amyloid aggregation or remodel preformed amylin8-24 fibrils is attenuated in the presence of negatively charged lipid bilayers [47,76]. These data suggest that key polar and non-polar regions of amylin that mediate the non-specific hydrophobic interactions and hydrogen bonding as suggested by Raleigh and colleagues [49], are sequestered by both lipid bilayers and detergent, leading to fewer interactions with EGCG.
Natural product-based amyloid inhibitors
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5841551/
There are many other natural compounds that show great promise in inhibiting amyloid formation. Resveratrol is one of them. I highly recommend reading the entire article that I have extensively quoted in this post. It is a wonderful find.
The heat wave has passed and I look forward to a seasonal summer weekend here in Vermont. One during which I plan to enjoy drinking alternating cups of Turmeric and Green Teas! Blessings, good health and thanks to all for your readership, dialog, support and encouragement.


God bless you, Walter❤️😊
TYVM Walt. Have a chill weekend.