Spike Protein Injury: The Foundational Basis for Post-Spike Protein Exposure Systemic Calcification
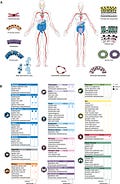
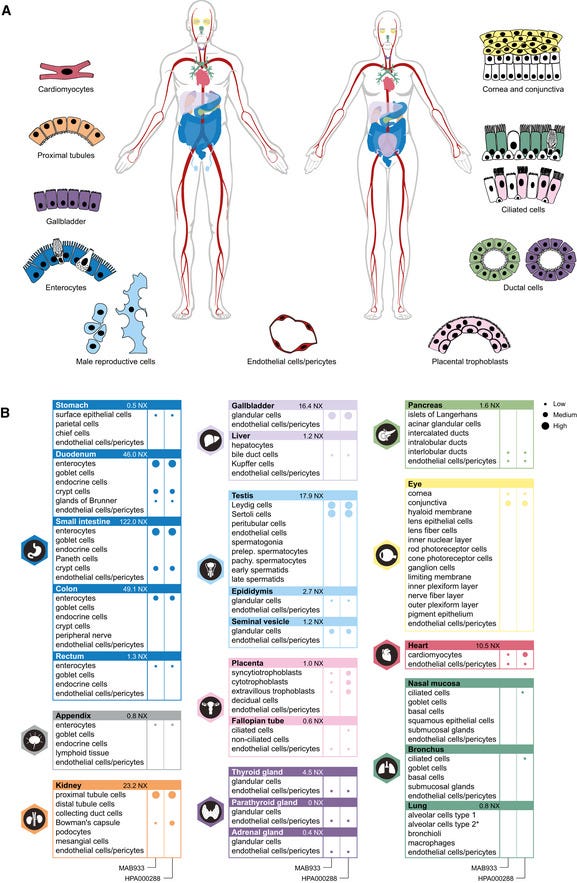
Why ACE2 as receptor is far more dangerous than most realize.
ACE2 protein expression in human tissues based on immunohistochemistry
My research has led me to conclude that the Spike Protein, in addition to many already known pathologies, is also inducing systemic calcification. To understand this it is first necessary to understand how incredibly dangerous having ACE2 as a receptor is to the anatomy of the human body. If you wanted to design a weapon to target as much of the human body as possible, you would be hard pressed to find a more viable candidate than ACE2. I do not believe this is an accident. Please see the above graphic.
Since the Spike Protein is cytotoxic, it causes injury to tissue. This is the foundation on which the induction of systemic calcification may occur:
As you can see, almost every calcification that one sees in the soft tissues in actual radiographic practice is due to dystrophic calcification. What does this mean? Simply this: when tissue is damaged, the body responds to this injury in a nonspecific manner by invoking the generic inflammatory response reaction. This sometimes ends with calcification of the damaged tissue. This calcification is probably usually only microscopic, but is occasionally enough to be seen radiographically.
Soft Tissue Calcifications
https://rad.washington.edu/about-us/academic-sections/musculoskeletal-radiology/teaching-materials/online-musculoskeletal-radiology-book/soft-tissue-calcifications/
Furthermore, there is an abundance of evidence that this is occurring post COVID or Spike Protein exposure.
BRAIN
Brain CT showing bilateral brain calcifications
In a recent manuscript published in Neurological Sciences, Demir (2020) attributed the incidental findings of bilateral brain calcifications in neuroimaging of a patient during SARS-CoV-2 infection to a condition known as Fahr’s syndrome. In this case, there was an important contribution, since neurological symptoms enabled the incidental diagnosis of a rare neurological disorder.
COVID-19 Unveiling Brain Calcifications
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8364776/
CORONARY
Results: The multivariate analysis proved that the OCS (Ordinal CAC Score) value was statistically correlated with the mortality rate (p < 0.001). In fact, in the group of patients with an OCS value of 0, the mortality rate was 10.1% (10/99 patients), in the group with OCS between 1 and 4 was 18.9% (21/111), in the OCS group of patients ranged from 5 to 8 was 30.4% (24/79) and in the OCS group between 9 and 12 was 46.4% (13/28).
Conclusions: We suggest that calcific atheromasia of the coronary arteries in patients with COVID-19 can be considered a prognostic marker of clinical outcome.
Coronary calcifications as a new prognostic marker in COVID-19 patients: role of CT
https://pubmed.ncbi.nlm.nih.gov/36930517/
PULMONARY
A 49-year-old man with Liddle syndrome type 1 with high blood pressure and chronic renal impairment, kidney transplant recipient, with hypoparathyroidism due to parathyroidectomy was admitted for bilateral COVID-19 pneumonia. Chest computed tomography (CT) performed at admission showed multiple foci of pulmonary calcifications coinciding with areas of opacities seen on a CT performed before admission, corresponding to foci of bronchopneumonia.
Pulmonary Calcifications as COVID-19 Pneumonia Sequelae
https://www.archbronconeumol.org/en-translated-article-pulmonary-calcifications-as-articulo-S0300289621004154
FETUS
A pregnant woman was diagnosed with SARS-CoV-2 at 35+5 weeks of gestation and managed conservatively at home. At transabdominal ultrasound at 38+3 weeks, fetal bowel and gallbladder calcifications were noted. CMV and other infectious agents were ruled out; an iterative caesarean section was performed at 38+5 weeks without complications.
Pregnant woman infected by Coronavirus disease (COVID-19) and calcifications of the fetal bowel and gallbladder.
https://www.unboundmedicine.com/medline/citation/33249821/Pregnant_woman_infected_by_Coronavirus_Disease_(COVID-19)_and_calcifications_of_the_fetal_bowel_and_gallbladder:_a_case_report.
MICROVASCULAR
While initial reports regarding coronavirus disease 2019 (COVID-19) focused on its pulmonary manifestations, more recent literature describes multisystem abnormalities related to its associated microvascular angiopathy. Calciphylaxis is a rare systemic condition characterized by tissue necrosis in the setting of systemic microvascular calcifications. Both COVID-19 and calciphylaxis are procoagulant diagnoses associated with vascular-mediated cutaneous findings. To our knowledge, this is the first report to document the coexistence of COVID-19 associated retiform thrombotic purpura and calciphylaxis in a single patient, to link the pathologic etiologies of the two entities, and to describe the concomitant diagnoses’ associated radiologic findings.
Concomitant calciphylaxis and COVID-19 associated thrombotic retiform purpura
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7447529/
While these are initial findings from research into multiple studies, I believe the case is clear. We must determine the extent of this phenomenon, as I believe it is likely to be extremely prevalent – simply not looked for to date. I will now research the progression of these conditions, and treatments for them.


Thank you ever so much for sharing your wisdom... speaking the truth, sharing facts & enlightening people. It is the most important thing we can do. The ones that have harmed our nation and the other global nations live in a culture of death and destruction. Only they should carry on, the rest of us are unnecessary and certainly in their selfish world there should be limits to any more people being born. So they’ve taken it into their own hands to bring on death and destruction....to give shots to pregnant women that will kill their babies...to give shots to little children, teenagers, young people harming them deliberately, and of course tell the older people that they must get a shot and boosters or you will die when in actuality, the more boosters you have, you will die because your immune system lessens each time. Once again, they lie...”Dear God please bring back truth and respect to our nation and our world....
Let the so called elites BEGONE!!
Thank you Walter! You are amazing!!!