Parallels of the Spike Protein and Hutchinson-Gilford Progeria Syndrome (HGPS) in Accelerated Aging
The Spike Protein mimics the effects of Progerin
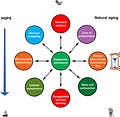
Nine hallmarks of aging (Lopez-Otın et al., 2013) contribute to both natural and premature aging. Epigenetic alterations are in a key position to affect all hallmarks of aging and to be affected by them. Mutual influences between other hallmarks that probably exist are not shown.
Everyone knows that a mutation in the Lamin A gene is the genetic cause of HGPS. However, it is not this mutation in and of itself which leads to the rapid aging in HGPS. It is the how having this mutation affects cellular processes which results in premature and rapid aging. What is important is that the Spike Protein, in essence, acts upon the same cellular processes as the mutated Lamin A gene.
Let us look at how the mutated gene causes the effects of accelerated aging. In particular, let us look at two examples. Cardiovascular and Nuclear/DNA damage.
CARDIOVASCULAR
Cardiovascular disorders are widely believed to be among the most characteristic diseases of advanced age. Similarly, such disorders are among the most typical features of the progerin-induced accelerated aging, probably due to the accumulation of progerin in arterial walls.
Also note:
Thus, abnormal accumulation of unprocessed pre-lamin A could induce DNA damage and mitotic dysfunction, eventually leading to cellular senescence. VSMCs are MSC-like cells that can undergo osteogenic and adipogenic differentiation, leading to calcification and lipid accumulation, pathologies prevalent in the vasculature of children with HGPS and transgenic mice overexpressing progerin. Pre-lamin A accumulation in VSMCs appears to be a causal factor of their senescence, since it precedes the senescence, and its overexpression accelerates senescence. Similar to progerin in HGPS fibroblasts, excessive pre-lamin A seems to be a dominant factor that disrupts nuclear lamina integrity, deregulates DNA damage response and mitotic checkpoints. In the context of cardiovascular disease, it could result in accelerated senescence of atherosclerotic plaques.
Are There Common Mechanisms Between the Hutchinson–Gilford Progeria Syndrome and Natural Aging?
https://www.frontiersin.org/articles/10.3389/fgene.2019.00455/full
This is precisely what the Spike Protein does. It mimics Progerin in the context of HGPS.
While the gold standard for detection of SARS-CoV-2 in tissue sections has yet to be determined, the detection of SARS-CoV-2 SP alone without spike RNA suggests that cleaved SP may be present in cutaneous endothelial cells and eccrine epithelium, providing a potential pathogenetic mechanism of COVID-19 endotheliitis.
Discordant anti-SARS-CoV-2 spike protein and RNA staining in cutaneous perniotic lesions suggests endothelial deposition of cleaved spike protein.
https://pesquisa.bvsalud.org/global-literature-on-novel-coronavirus-2019-ncov/resource/pt/covidwho-748637
NUCLEAR DAMAGE
In HGPS the effects of the mutated Lamin A gene affects the nuclear membrane and induces DNA damage.
The mutant nuclear lamin protein (progerin) produced in Hutchinson-Gilford progeria syndrome (HGPS) results in loss of arterial smooth muscle cells (SMCs), but the mechanism has been unclear. We found that progerin induces repetitive nuclear membrane (NM) ruptures, DNA damage, and cell death in cultured SMCs.
Nuclear membrane ruptures underlie the vascular pathology in a mouse model of Hutchinson-Gilford progeria syndrome
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8409987/
And, yes, the Spike Protein induces the very same effects.
Interaction between the S protein and ACE2 can not only mediate virus entry, but also induce cell-cell fusion, resulting in the formation of syncytia - cells with multiple nuclei. Nuclei in the fused cells have lower levels of lamin A/C, the major contributor of the mechanical property of the nucleus, and have evident nuclear membrane blebs containing DNA. These blebs protrude from the nuclear lamina and rupture, leading to the release of DNA to the cytoplasm. Consistently, DNA damage foci, stained by γH2AX, are found to accumulate in infected cells with lower lamin A/C. diABZI, a potent STING activator, greatly reduces SARS-CoV-2 infection. These results suggest that nuclear integrity modulates host cell immune response during virus infection.
SARS-CoV-2 and the Nucleus
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9305274/
In addition to the Spike mimicking the effects of radiation, most likely inducing fibrosis (see previous post), the Spike most likely also mimics the effects of Progerin, causing pathologies parallel to HGPS.


That genome instability one interests me. Integration of small foreign bits of DNA can lead to genome instability. R-loops that accumulate due to a reduced ability to degrade DNA:RNA hybrids also cause genome instability and can lead to DNA damage.
Long Vax syndrome
Is difficult to distinguish from Long Covid
Both presenting with extreme fatigue, cognitive difficulties and post exertional malaise (PEM) amongst other symptoms
And mitochondrial dysfunction seems to underpin both along with a number of other melodies with similar presentations.
Such as Gulf war syndrome
Chronic Lyme disease
CFS/ME
And the side effects of flueroquinolone antibiotics commonly referred to as Floxed
Not good
Crippling
Sickling
And slowly sapping morale, vigor and any ability to run or retaliate
A particularly effective form of incapacitation
To help multinationals steal your nation
From right under your nose