Multiple Study Results Demonstrate COVID May Induce a Novel Delayed Brain Disorder Similar to SSPE
Imaging and longitudinal studies show that Long COVID results in similar conditions to Subacute Sclerosing Panencephalitis (SSPE).
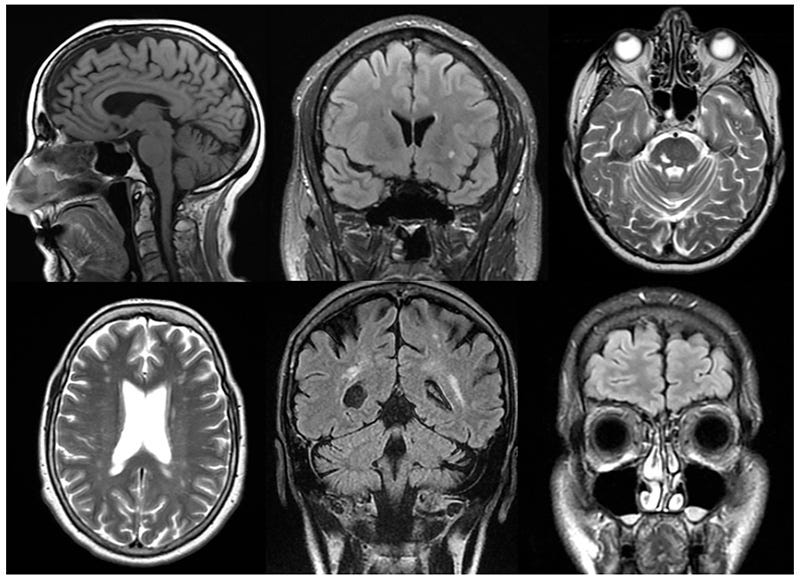
From left to right in the first row are cortical atrophy, white matter lesion, and vascular lesions and, in the second row, are lacunar lesion, vascular encephalopathy, and sinusitis.
It was four years ago that I wrote about a mechanism that I saw could be shared between Measles and COVID. With Measles, some individuals experience neuroinvasion which can lead to a form of viral persistence in which its N protein causes a fatal neurological disease, years later.
Subacute sclerosing panencephalitis (SSPE) is thought to be a long-term measles virus infection. The virus sometimes enters the brain during a measles infection. Measles virus may cause immediate symptoms of brain infection (encephalitis), or the virus may remain in the brain for a long time without causing problems.
Subacute Sclerosing Panencephalitis (SSPE)
https://www.merckmanuals.com/home/children-s-health-issues/common-viral-infections-in-infants-and-children/subacute-sclerosing-panencephalitis-sspe
Given that far, far more individuals experience COVID neuroinvasion, the parallel greatly concerned (and still concerns) me.
A DARK STORM GATHERING: DISTURBING PARALLELS BETWEEN THE MEASLES N PROTEIN AND THE SARS-CoV-2 S PROTEIN: SUBACUTE SCLEROSING PANENCEPHALITIS
https://wmcresearch.substack.com/p/a-dark-storm-gathering-disturbing
Now, years later, we have the results of several studies which confirm that long-term damage similar to SSPE is being caused by Long COVID. Let me be clear; I am not stating that it IS SSPE. I am stating that COVID may induce a delayed, persistent viral encephalitis analogous to Subacute Sclerosing Panencephalitis.
First, let’s look at imaging results.
In SSPE, lesions of high signal intensity on T2-weighted images are the most common finding; they frequently involve the periventricular or subcortical white matter.
Thirty-four MRI studies of 26 patients with subacute sclerosing panencephalitis are reported. Lesions of high signal intensity on T2-weighted images are the most common finding; they frequently involve the periventricular or subcortical white matter. Lesions tend to start in the cortex-subcortical white matter and progress with periventricular white matter involvement and diffuse cerebral atrophy. Pial and parenchymal contrast enhancement, local mass effect of parenchymal lesions, and involvement of the splenic portion of the corpus callosum are not infrequent.
MRI findings in subacute sclerosing panencephalitis
https://pubmed.ncbi.nlm.nih.gov/8909443/
Note, again, that they occur in the periventricular and subcortical white matter. This is also the case in Long COVID.
White matter lesions: the Fazekas scale and the Standards for Reporting Vascular Changes on Neuroimaging (STRIVE) criteria are the most commonly used standards. White matter lesions are identified on FLAIR and T2-weighted images as bright (hyperintense) areas located in the periventricular or deep white matter.
Diffuse white matter hyperintensities are observed on FLAIR images. Subcortical infarcts, dilated perivascular spaces, or signs of microvascular disease may also be present. Cortical and subcortical atrophy is frequently associated with ventricular enlargement [17].
Brain Structural Abnormalities in Patients with Post-COVID-19 Headache
https://www.mdpi.com/2035-8377/17/4/50
Another aspect of SSPE is the presence of demyelination and neurofibrillary tangles.
Early stages of CNS infection cause cellular swelling and oxidative damage to genetic material. It is thought that lipid peroxidation causes demyelination in the early course of the disease. This is followed by an acute inflammatory phase where nucleocapsids are evident in oligodendrocytes and neurons, while granulofilamentous inclusions within nuclear bodies are present in astrocytes. Inflammatory cells can be found in perivascular regions along with scattered areas of demyelination and spongiosis.[4][5][8] Neurofibrillary tangles have been found in some cases of SSPE.[9]
Subacute Sclerosing Panencephalitis
https://www.ncbi.nlm.nih.gov/books/NBK560673/
A Swedish study showed that there is, indeed, a marked increase in the occurrence of demyelinating diseases post COVID.
Hospital admission for COVID-19 was associated with raised risk of subsequent non-multiple sclerosis demyelinating disease, but only 12 individuals had this outcome among the exposed, and of those, 7 has an unspecified demyelinating disease diagnosis. Rates per 100 000 person-years (and 95% confidence intervals) were 3.8 (3.6–4.1) among those without a COVID-19 diagnosis and 9.0 (5.1–15.9) among those admitted to hospital for COVID-19, with an adjusted hazard ratio and (and 95% confidence interval) of 2.35 (1.32–4.18, P = 0.004). Equivalent associations with multiple sclerosis (28 individuals had this outcome among the exposed) were rates of 9.5 (9.1–9.9) and 21.0 (14.5–30.5) and an adjusted hazard ratio of 2.48 (1.70–3.61, P < 0.001).
SARS-CoV-2 infection and risk of subsequent demyelinating diseases: national register–based cohort study
https://academic.oup.com/braincomms/article/6/6/fcae406/7909395
A just-published study shows the relation between Long COVID and the potential development (definite presence of amyloid) of neurofibrillary tangles.
Our study found that patients who develop N-PASC after COVID-19 might share certain clinicopathological features with AD. Indeed, while Tau functions adaptively to stabilise neuronal microtubules under normal physiologic conditions23 and can also be dysregulated and spread by reactive glia,41 especially in the context of chronic inflammation.42 However, the prognostic implications of increases in circulating pTau-181 absent concurrent amyloidosis are unknown. Usually, β-amyloid peptide plays a central role in triggering Tau phosphorylation in ADRD,43 a process that leads to subsequent microtubule destabilisation resulting in threads that coalesce into neuronal tangles.44 Intriguingly, in this study we found that evidence of increased pTau-181 was associated with increases in AB40/42 ratios consistent with a pTau-mediated ADRD. Further studies are needed to determine whether the increased levels of plasma pTau-181 correlate with evidence of cerebral Tauopathy and, if results are replicated, pTau-181 might aid in diagnosis and might serve as an important monitoring and therapeutic target.45
We found that individuals who developed N-PASC had higher Aβ40/42 ratios (AUC = 0.63, P = 0.007), NfL (AUC = 0.59, P = 0.002), and IAB values (AUC = 0.74, P < 0.001) before developing COVID-19. Higher values suggest that N-PASC might be more likely in those individuals who have heightened vulnerability to neurological disease. Amyloidosis often requires a secondary neuropathology to elicit the most severe symptomatology. If these findings indicate that cerebral amyloidosis is present, even in its mildest forms, then the post-COVID-19 increase in pTau-181 may correspond to the onset of pathological Alzheimer’s disease.
Increased phosphorylated tau (pTau-181) is associated with neurological post-acute sequelae of coronavirus disease in essential workers: a prospective cohort study before and after COVID-19 onset
https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(25)00556-0/fulltext
So, we have evidence that COVID may be inducing a potentially very harmful long term CNS disorder. Why this concerns me far more than the SSPE complication of Measles is something that has concerned me from Day One: Reinfections. Measles infection almost always results in lifelong immunity. COVID infection can reoccur within weeks – and regularly. I will continue to investigate this phenomenon, seek therapeutics, and continue to report back to you. Please have a blessed week.

Isoprinosine (also known as Inosine Pranobex or Methisoprinol) is
an antiviral and immunomodulatory agent commonly used in combination with interferon-alpha to treat Subacute Sclerosing Panencephalitis (SSPE). While SSPE is a progressive and usually fatal neurodegenerative disease caused by the measles virus, treatment with Isoprinosine has shown potential to stabilize the disease, improve symptoms, and significantly prolong survival.
Key Findings on Isoprinosine for SSPE:
Effectiveness: Studies indicate that Isoprinosine can lead to long-term remission in about 33–35% of cases, which is significantly better than the 5% spontaneous remission rate in untreated cases.
Survival Rates: One study noted an 8-year survival rate of 61% in patients treated with Isoprinosine compared to only 8% in untreated patients.
Combination Therapy: Combining oral Isoprinosine with intraventricular interferon-alpha is considered the most effective treatment, potentially leading to stabilization or improvement in 44–55% of cases.
Administration: It is typically administered orally at doses of 50–100 mg/kg/day.
Side Effects: The drug is generally well-tolerated, with occasional reports of mild hyperuricemia (elevated uric acid in the blood and urine).
Limitations: While it can slow or stabilize progression, it often cannot fully reverse damage already caused by the disease.
Thanks Walter. I appreciate the papers and information that seem to illustrate what I am seeing.